我的思考
注意,非等价掺杂引入的缺陷可能导致格位对称性发生变化:
- These defects can produce Ln3+ sites with distinct symmetries, depending on their position relative to the dopant. (Optical Materials, 2022, 133: 112926.)
-
Unusual Concentration Induced Antithermal Quenching of the Bi2+ Emission from Sr2P2O7:Bi2+
Li4SrCa(SiO4)2
禁带宽度:The band F at 167 nm is assigned to the host excitonic absorption, from which the host band gap is estimated to be 8.02 eV after taking into account the electron–hole binding energy of the exciton. (参考Rui Shi-IC-2018)
变化合成条件——调整Eu的占位:
- 固溶体调控;
- Eu的掺杂浓度很重要,如果有浓度高了,很容易有不同格位的能量传递,这也是为什么有的人观察到ET,有的人没有观察到。我选择0.004左右的掺杂浓度。
- 不同的合成温度,比如A highly efficient green-emitting phosphor, (Na0.97Eu0.03)BaB9O15 was successfully synthesized by aging the starting materials for 2 weeks and lowering the sintering temperature to drive the substitution of Eu2+ onto the smaller, less favorable [NaO6] substitution site. (参考CM-Jakoah Brgoch-2021)
- 变化合成温度,比如参考K2Ca(PO4)F:Eu2+。
- 是不是高对称性的更容易光还原?
- 格位占位分析,可以参考ACS Appl. Mater. Interfaces 18, 11615 (2016)和J. Appl. Phys. 129, 200903 (2021)
- 是否可以采用低温氢气气氛热处理,让被光还原的Eu2+冻住,这样就可以更好地用于温度传感。注意此处热处理的温度不能超过Eu3+样品,低温还原气氛热处理得到Eu2+的温度。
参考文献,La2Zr2O7,500度热处理的时候,Eu3+处在样品的表面,而到了700度,就进入了La3+的格位。 - 文献BaMgSiO4-Eu2+先在高温下合成粉末(1000度,空气),然后压成片在高温下处理(1350度,还原气氛),然后分别在1000/1100/1300度下处理一个小时(空气),最后测PL发现Eu2+的占位发生了变化。
- Eu2+配位数小的,发长波长光,参考Rb3YSi2O7:Eu
Eu3+格位对称性:
- 电偶极跃迁,即便是Eu3+所处的格位仅稍微偏离反演对称性,此跃迁发射在光谱中也占主导地位。(固体发光材料-P89)
- Excitation-dependent local symmetry reversal in single host lattice Ba2A(BO3)2:Eu3+ [A = Mg and Ca] phosphors with tunable emission colours
测试:
- 测Sr格位的发光要避免激发Eu3+以及Ca格位;
- 测Ca格位的发光要避免激发Eu3+以及Sr格位;
Sr-rich橙色格位的非对称性
- A special feature of SALON:Eu2+ is the slightly asymmetric emission band at room temperature. A number of theoretical studies have been published on SALON:Eu2+ to explain both the emission wavelength and asymmetry of this emission band. [来自Markus Suta-AOM-2023]
热稳定性:
- 参考固体发光材料—P130,Blasse总结了碱土金属荧光材料的猝灭温度通常按照Ca/Sr/Ba的顺序逐渐升高。
应用:可能可以作为温度传感,或者X射线剂量。
其他有意思的基质:
- K2BaCa(PO4)2−x(BO3)x:Eu,这篇文章列举了各种双峰Eu2+发射。Eu3+的荧光粉,辐照一下,可能都跑到发红色宽带发光的格位,然后低温还原气氛处理,可能得到的就是比较纯的红色荧光粉。
- K2Ca(PO4)F:Eu2+ Eu3+应该不太容易占据K+的格位。
- K3YSi2O7,Ce3+掺杂只占一个格位,Eu2+占据两个。
- NaBa3Si2O7F,两个格位
- Multi-site occupancies of Eu2+ in Ca6BaP4O17 and their potential optical thermometric applications
Eu2+掺杂
非固溶体
1.[The New Orthosilicate Li4SrCa(SiO4)2: Structure and Eu2+ Luminescence-IC-1995]
制备方法:The mixture of carbonates and Si02 was heated at 873 K for 1 h; then the sample was reground and heated at 1273 K for 12 h.

结构和发光:
- a 10-coordinate Sr atom, a 6-coordinate Ca atom;
- the Sr atom is best described as a distorted bicapped square antiprism, and the polyhedron about the Ca atom is a distorted octahedron.
- Ca具有C2 symmetry。
- 和典型的硅酸盐,比如Sr2SiO4 (550 nm),Ca2SiO4 (500 nm)相比,这里的发光峰波长更短。The higher emission energy of the title compound may derive from the high concentration of Li atoms in the structure. These small atoms, surrounding the polyhedra of the emission centers, likely constrain the distortion of the excited state which leads to a smaller Stokes shift.
上面两篇是同一个人发表的。
制备方法:
- air at 600 °C for 1 h, and re-sintered in reducing atmosphere at 900 °C for 12 h.
- thoroughly mixed and then sintered in air at 600°C for 1 h, and resintered at 1000°C for 12 h.
光学性质:body color is greyish white; 热猝灭的激活能为0.35 eV
类似组分的荧光粉对比:

3. [Li2Ca0.7Sr0.3SiO4:Eu2+ and its application—JAC-2010]
样品制备:i2CO3, CaCO3, SrCO3, SiO2, together with 10% in molar ratio of extra Li2CO3, were mixed thoroughly in alcohol by ball milling in agate mortar and then dried in an oven. The mixed powders were firstly pre-fired at 600 °C for 1 h in air. And then the final synthesis was carried out at 900 °C in a reductive atmosphere composed of 90% N2 and 10% H2。Eu2+的掺杂浓度也是1%(作者说此浓度发光最强,但是没有给出具体结果)。

光学性质:
- The experimental results show that the emission intensity is little decreased after baking in atmosphere at 80 °C for 5 h.
- 465 nm的峰可能是少量的杂相的峰;
- 作者对于Eu2+占位的分析基于以下理由,但是是结论是错误的:
- the Ca2+ and Sr2+ are both divalent which share the same amount of electrons;
- the ion radius of Ca2+ is smaller than Sr2+ which results in weaker crystal field strength;
- the average bong length of Sr–O 2.76 ± 0.16 Å is larger than that of Ca–O 2.384 ± 0.026 Å
- the coordination number of Ca2+ is less than that of Sr2+ so that the electron cloud expansion effect is weaker which results in higher energy of the excited level of Eu2+.
6. [兰州大学-李叶洲-博士论文]
稀土掺杂的硅酸盐由于具有丰富的晶体结构,很好的物理和化学稳定性,以及优越的发光性能受到广泛关注。该结构中含有SrO10多面体 畸变的 \(\mathrm{CaO}_6\) 八面体, 畸变的 \(\mathrm{LiO}_4\) 四面体和 \(\mathrm{SiO}_4\) 四面体, 其中 \(\mathrm{Sr}\) 原子 周围的 \(\mathrm{O}\) 原子分布可以描述为一畸变的双帽直角反棱柱结构, 这一结构很难用 最密球堆积来形容, 但是可以肯定的是其中的金属离子为层状堆垛结构, 即 \(\mathrm{Ca}\) 和 \(\mathrm{Si} 2\) 原子为一层, \(\mathrm{Li}\) 和 \(\mathrm{Li} 2\) 原子为一层, \(\mathrm{Sr}\) 和 \(\mathrm{Si} 1\) 原子为一层。当稀土离子进 入 \(\mathrm{Li}_4 \mathrm{SrCa}\left(\mathrm{SiO}_4\right)_2\) 基质时, 可能取代 \(\mathrm{Sr}^{2+}\) 或者 \(\mathrm{Ca}^{2+}\) 的位置, 而它们各自与同类原 子之间的能量传递可以被密堆的层状结构所有效阻隔, 因此当掺杂浓度增加时不 易发生浓度猝灭, 有可能产生较高的发光强度。另外由于 \(\mathrm{SiO}_4\) 四面体在 VUV 区 域内有吸收, 所以稀土激活的 \(\mathrm{Li}_4 \mathrm{SrCa}\left(\mathrm{SiO}_4\right)_2\) 也很有可能在 \(\mathrm{VUV}\) 光子的激发下 产生发光。
图5-3为合成的 \(\mathrm{Li}_4 \mathrm{SrCa}\left(\mathrm{SiO}_4\right)_2: \mathrm{Eu}^{2+}\) 样品的晶胞参数 \(a 、 b 、 c\) 以及晶胞体积 \(V\) 随 \(\mathrm{Eu}^{2+}\) 掺入量增加的变化趋势图。由图可见: 晶胞参数 \(a\) 随着 \(\mathrm{Eu}^{2+}\) 浓度的增加基本 没有发生明显的变化, 而 \(b 、 c\) 以及晶胞体积 \(V\) 与随着 \(\mathrm{Eu}^{2+}\) 浓度的增加呈现出线性 递减的趋势, 说明了 \(\mathrm{Eu}^{2+}\) 的掺杂主要影响 LSCSO晶胞中 \(b\) 和 \(c\) 轴方向的大小。通过 在图5-1LSCSO晶体结构的分析, 可以知道 \(\mathrm{Ca}^{2+}\) 是严格沿着b轴和c轴方向分布的, 而 \(\mathrm{Sr}^{2+}\) 只是严格沿着b轴方向分布。因此, 系列样品晶胞参数的变化结果, 尤其是 c值的变化趋势说明在LSCSO中, \(\mathrm{Eu}^{2+}\) 取代的应该是 \(\mathrm{Ca}^{2+}\) 格位。另外, 之所以它 们的 \(b 、 c\) 以及晶胞体积 \(V\) 随着 \(\mathrm{Eu}^{2+}\) 浓度增加而呈现线性减小的变化, 这是由于 \(\mathrm{Eu}^{2+}\) \((1.06 \AA)\) 的离子半径比 \(\mathrm{Ca}^{2+}(1.10 \AA)\) 小, 这种线性变化符合Vegard定则 \({ }^{[7-9]}\), 说明 在掺入 \(\mathrm{Eu}^{2+}\) 的系列样品中 \(\mathrm{Eu}^{2+}\) 都进入了基质晶格并取代了 \(\mathrm{Ca}^{2+}\) 的位置而形成发光 中心。
 系列样品的发射光谱。由图可以看出, \(\mathrm{Li}_4 \mathrm{SrCa}\left(\mathrm{SiO}_4\right)_2: \mathrm{Eu}^{2+}\) 在该近紫外光激发下 可以产生较强的宽带发射, 该发射光谱由一个位于 \(432 \mathrm{~nm}\) 处占主导地位的宽峰、 在 525 675 nm 范围内的弱带以及叠加在上面的数个较弱的尖峰构成。其中, 强、 弱两个宽峰可以分别归属为 \(\mathrm{Eu}^{2+}\) 占据两种格位所产生的特征发射 \({ }^{[16-17]}\) 。根据前面 的结果, 可以知道 \(\mathrm{Eu}^{2+}\) 倾向于占据 \(\mathrm{Ca}^{2+}\) 的位置, 因此位于 \(432 \mathrm{~nm}\) 处的主峰应该 归属为 \(\mathrm{Eu}^{2+}\) 占据 \(\mathrm{Ca}\) 位置所产生的发射; 而之后较弱的宽峰归属为 \(\mathrm{Eu}^{2+}\) 占据 \(\mathrm{Sr}\) 位置所产生的发射。数个较弱的尖峰可以归属为 \(\mathrm{Eu}^{3+}{ }^{3+} 子\) 离的特征 \(\mathrm{f}-\mathrm{f}\) 跃迁。另外, 我们通过实验可知 \(\mathrm{Li}_4 \mathrm{SrCa}\left(\mathrm{SiO}_4\right)_2: \mathrm{Eu}^{2+}\) 中不会发生自还原, 因而会产生 \(\mathrm{Eu}^{3+}\) 的发 光的原因, 我们认为有以下几点: 从晶体结构上看, 10 配位的 \(\mathrm{Sr}\) 多面体为层状 分布, 而 \(\mathrm{Ca}\) 六面体之间则是被隔离的 \({ }^{[6]}\), 换句话说, \(\mathrm{Ca}\) 位置是被多个多面体形 成的网状结构严密地包围起来的, 因此在还原气氛下, 占据了 \(\mathrm{Ca}\) 位置的那部分 \(\mathrm{Eu}^{3+}\) 离子有可能被网状结构所屏蔽而不能被有效还原, 因此产生了一些较弱的 \(\mathrm{Eu}^{3+}\) 的特征发射, 在光谱上表现为一些强度很弱的小尖峰。
系列样品的发射光谱。由图可以看出, \(\mathrm{Li}_4 \mathrm{SrCa}\left(\mathrm{SiO}_4\right)_2: \mathrm{Eu}^{2+}\) 在该近紫外光激发下 可以产生较强的宽带发射, 该发射光谱由一个位于 \(432 \mathrm{~nm}\) 处占主导地位的宽峰、 在 525 675 nm 范围内的弱带以及叠加在上面的数个较弱的尖峰构成。其中, 强、 弱两个宽峰可以分别归属为 \(\mathrm{Eu}^{2+}\) 占据两种格位所产生的特征发射 \({ }^{[16-17]}\) 。根据前面 的结果, 可以知道 \(\mathrm{Eu}^{2+}\) 倾向于占据 \(\mathrm{Ca}^{2+}\) 的位置, 因此位于 \(432 \mathrm{~nm}\) 处的主峰应该 归属为 \(\mathrm{Eu}^{2+}\) 占据 \(\mathrm{Ca}\) 位置所产生的发射; 而之后较弱的宽峰归属为 \(\mathrm{Eu}^{2+}\) 占据 \(\mathrm{Sr}\) 位置所产生的发射。数个较弱的尖峰可以归属为 \(\mathrm{Eu}^{3+}{ }^{3+} 子\) 离的特征 \(\mathrm{f}-\mathrm{f}\) 跃迁。另外, 我们通过实验可知 \(\mathrm{Li}_4 \mathrm{SrCa}\left(\mathrm{SiO}_4\right)_2: \mathrm{Eu}^{2+}\) 中不会发生自还原, 因而会产生 \(\mathrm{Eu}^{3+}\) 的发 光的原因, 我们认为有以下几点: 从晶体结构上看, 10 配位的 \(\mathrm{Sr}\) 多面体为层状 分布, 而 \(\mathrm{Ca}\) 六面体之间则是被隔离的 \({ }^{[6]}\), 换句话说, \(\mathrm{Ca}\) 位置是被多个多面体形 成的网状结构严密地包围起来的, 因此在还原气氛下, 占据了 \(\mathrm{Ca}\) 位置的那部分 \(\mathrm{Eu}^{3+}\) 离子有可能被网状结构所屏蔽而不能被有效还原, 因此产生了一些较弱的 \(\mathrm{Eu}^{3+}\) 的特征发射, 在光谱上表现为一些强度很弱的小尖峰。
固溶体
制备方法:Stoichiometric amounts of the starting reagents were thoroughly mixed and ground together by an agate mortar. The mixture was then calcined at 800°C for 8 h in a reduction atmosphere (N2: H2 = 95: 5).
光学性质:

- 通过K-M方法得到的光学禁带宽度为5.1 eV;
- 注意这里的掺杂浓度为4 mol%,可能导致更为显著的ET;
- 激发谱:
- 监测蓝光:299 nm;
- 监测黄光:258 nm,299 nm,385 nm,相对强弱和x有关。
- 作者认为蓝光峰对应于Sr2+格位,而黄光峰对应于Ca2+格位。
- 第二张图,表面存在Eu3+,即还原不完全。
- 第四张图,浓度猝灭:
- x大于0.3之后,Ca2+格位的发光也在减弱,可能发生了浓度猝灭;
- x小于-0.2之后,Sr2+格位的发光也在减弱,可能发生了浓度猝灭
- 量子效率:
- The external QE of the typical Li4Ca0.8Sr1.16(SiO4)2:0.04Eu2+ sample was measured to be about 14.8%.
- The external QE of the typical Li4Ca1.2Sr0.76(SiO4)2:0.04Eu2+ sample was measured to be about 22.1%.
- The temperature-dependent photoluminescence spectra reveal that the Eu2+ emission centers in the Sr2+ and Ca2+ sites have different thermal stabilities, owing to their different chemical environments.
Eu3+掺杂
1.[Li4SrCa(SiO4)2∶Eu3+红色荧光粉的合成及其发光性能]
制备方法:在空气气氛下, 先将原料低温(600 ℃)预烧 24 h,得到前驱体;冷却 后,再将前驱体研磨,在高温(900 ℃)煅烧,并在此温 度下保温 48 h,随后随炉冷却。

Ce3+掺杂
其他
Bi3+/Pb2+掺杂
1.[JL-2015]
制备方法:And then, they were mixed uniformly in an agate mortar and placed in a furnace for pre-calcination at 450 C for 1 h. After that, the powder samples removed from the furnace, and again mixed homogenously. Finally, the samples were fired at 1000 °C for 12 h in air.
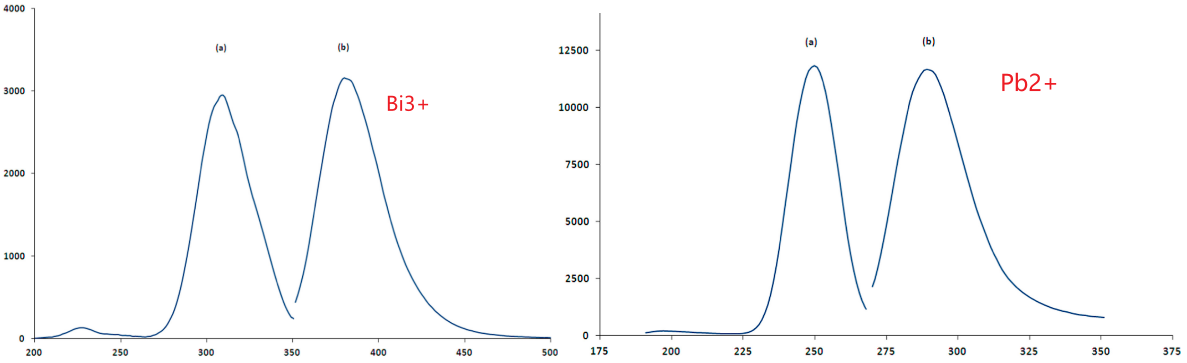
发光:
- 作者认为,两种掺杂离子都是只占据Sr格位,一种。
- 基质不发光。