Eu2þ/Eu3þ reduction and Tb3þ/Tb4þ transformation in SrAl2Si2O8:Eu/Tb system
McKeever, S. W. S., et al. Radiation Measurements 133 (2020): 106278.
Observation of Eu2+ in Y2O2S:Eu3+ and Y2O2S:Eu3+, Tb3+
Laser Photochemistry of Lanthanide Ions
Photoreduction of Ytterbium and Samarium
我的总结
- 对于Eu3+的光致变价,可以从所占据的格位分类,
-
- 三价格位:如果Eu3+之前占据的是La/Y/Gd/Lu的三价,那么即使被辐照还原为Eu2+,由于Eu2+在三价格位发光很弱(甚至没有),可能需要在很低的温度下才能被观察到,所以可能被很多人忽略,也就是说没有观察到Eu2+很可能并不意味着没有Eu2+。
(1) [Photobleaching of LnVO4:Eu3+ nanoparticles under UV-light irradiation: Effect of nanoparticle size-JL-2022] 没有观察Eu2+,但是别人[Single europium-doped nanoparticles measure temporal pattern of reactive oxygen species production inside cells-Nature-Nanotech-2009]观察到了。
(2) [Fine structure in high resolution 4f7–4f65d excitation and emission spectra of X-ray induced Eu2+ centers in LuPO4: Eu sintered ceramics-JL-2019]观察到了Eu2+的发光,但是看起来很多毛刺,应该是发光很弱。
(3) [Photoluminescence and gamma-ray irradiation of SrAl2O4:Eu2+ and Y2O3:Eu3+ phosphors-JPCS-2007]中没有观察到Y2O3:Eu3+中通过光还原得到的Eu2+,只是从PL的角度没看到,但是[Effect of X-Ray Radiation on the Valence State of Europium in Y2O3:Eu3+ Phosphor- Physics of the Solid State-2019]从EPR信号中观察到了Eu2+。 - 二价格位:如果Eu3+之前占据的是三价或者一价离子的格位,那么辐照还原得到的Eu2+发光就很容易被观察到。
- 三价格位:如果Eu3+之前占据的是La/Y/Gd/Lu的三价,那么即使被辐照还原为Eu2+,由于Eu2+在三价格位发光很弱(甚至没有),可能需要在很低的温度下才能被观察到,所以可能被很多人忽略,也就是说没有观察到Eu2+很可能并不意味着没有Eu2+。
-
- 辐照后得到的Eu2+,在一定气氛和温度下annealing,可能得到stable的Eu2+的发光。比如文献[One-Electron Reduction of Eu3+ Ions Induced by the Irradiation of γ-Ray or UV-Light. The Fluorescence Properties of Eu2+ Ions in an Ethanol Matrix-Chemistry Letters-1988]说的These results suggest that the Eu2+ ions produced by γ-irradiation in a 77 K matrix were fixed in rather unstable states which make the fluorescence process inefficient. Upon the annealing, the Eu2+ ion relaxes to stable conditions and becomes to indicates normal fluorescence properties.
- Eu3+的发光比例很低(相对于Eu2+,即使采用有利于Eu3+的激发波长),并不意味着Eu3+不存在,XAS测试可能表明还存在相当数量的Eu3+。A considerable fraction of Eu3+ ions can be present in the powders, although this is not necessarily reflected in the photoluminescence properties.[Valence states of europium in CaAl2O4:Eu phosphors-OME-2012] In addition, phosphors showing only Eu2+ emission can still contain a relatively large fraction of Eu3+ ions. [Extending the afterglow in CaAl2O4:Eu,Nd persistent phosphors by electron beam annealing-OME-2012]
- 光致还原得到的Eu2+,很难完全褪掉:It is very difficult to bleach the last 1-2% of Sm2+ ions. The residual Sm2+ may be due to the temperature induced motion of the defect pairs which may lead to larger separations which are inaccessible for the electron transfer. [Highly efficient valence state switching of samarium in BaFCl:Sm nanocrystals in the deep UV for multilevel optical data storage] 这个和OSL类似:Filling of deep traps during high dose irradiation can create some difficulties when performing complete optical bleaching and may require high temperature annealing to completely zero the detector and reduce the background OSL signal caused by so-called “dark transfer” or leakage from the deep traps. These dark transfer processes are well known in TL materials. [Fundamentals of Materials, Techniques, and Instrumentation for OSL and FNTD Dosimetry]
- 完全还原的例子:Sm3+ ions can be completely reduced into Sm2+ by β-irradiation with doses higher than 109 Gy. [Optical properties and valence state of Sm ions in aluminoborosilicate glass under β-irradiation-JNCS-2007]
- 用于Laser-Lighting的Eu2+掺杂荧光粉,在高功率/高温下的变价(被氧化为Eu3+)[Reliability of Blue-Emitting Eu2+-Doped Phosphors for Laser-Lighting Applications]
- Owing to their reducing character, it is not surprising that Ln2+ ions are easily photooxidized. In particular, the photoreactivity of Eu(II) and Sm(II) compounds has been studied in some detail. In analogy to Ce(III) (see above), the photooxidations of Ln(II) seem to originate from fd states since the fd transitions terminate at d orbitals which are exposed to the ligands or the solvent. [Excited state properties of lanthanide complexes: Beyond ff states]
价态的表征
The determination of dopant ion valence distributions in insulating crystals using XANES measurements
飞秒激光系列(中国/日本)
1. Eu3+掺杂fluorozirconate玻璃
[Infrared femtosecond laser pulse–induced permanent reduction of Eu3+ to Eu2+ in a fluorozirconate glass-OL-1999][mathjax]
摘要:We report what is believed to be the first observation of permanent photoreduction of Eu3+ to Eu2+ in transparent and colorless Eu3+-doped fluorozirconate glass at room temperature, using an infrared femtosecond laser. Difference absorption and electron-spin-resonance spectra of the glass before and after laser irradiation showed that a portion of the Eu3+ ions in the focused part of the laser inside the glass were reduced to Eu2+ ions after laser irradiation. It is suggested that Eu3+ ions act as electron-trapping centers, whereas active sites in the glass matrix act as hole-trapping centers, leading to the formation of Eu2+ ions. The observed phenomenon is inferred to be useful in the fabrication of optical memory devices with high storage density and waveguide-type micro-optical devices.
激光的牛逼应用:lasers have been used as powerful tools to clarify elementary processes, such as excitation-energy relaxation and both electron and proton transfer on the nanosecond and picosecond time scales, that occur in a micrometer-sized area. Chemical-reaction mechanisms have also been elucidated for hydrogenatom transfer, isomerization reaction, and consecutive bond breaking by means of time-resolved laser spectroscopy.
用飞秒激光来对透明物质进行microscopic modifications的原因:the strength of its electric field can reach 100 TW/cm2, which is sufficient for inducing nonlinear optical effects in materials by use of a focusing lens, when the pulse width is 100 fs and the pulse energy is 1 mJ.
机理解释:飞秒激光用的是800 nm的光,但是样no absorption in the wavelength region near 800 nm, and therefore photoreduction of Eu3+ to Eu2+ should be a nonlinear optical process。激光辐照的时候也观察到了Eu3+的发光,其发射波长小于激发光的,因此multiphoton absorption occurred during laser irradiation。
变价过程:Active electrons and holes are created in the glass through a multiphoton ionization process. Holes are trapped in the active sites in the glass matrix, and some electrons are trapped by Eu3+, leading to the formation of V-type hole-trapped defect centers and Eu2+. Efimov et al. suggested that defect centers can be induced by the short-wavelength part of a supercontinuum that is due to self-phase modulation of an ultrashort pulsed laser.
2. Eu3+掺杂Al2O3−B2O3−SiO2玻璃
[Optical Properties and Valence Change of Europium Ions in a Sol−Gel Al2O3−B2O3−SiO2 Glass by Femtosecond Laser Pulses-JPCB-2004]
主要内容:
(1) the Eu3+ ions at sites with higher covalence degree can be more easily reduced than those at sites with lower covalence degree via femtosecond laser pulses. 通过Fluorescence line-narrowing spectra得到证实(激发源单色可调激光、8 K温度)。
(2) 辐照前后吸收峰变化,200 to 410 nm处的增强来自Eu2+的吸收,410 to 500 nm处的增强来自positive hole centers。后者的归属是基于别人文献报道的the gamma irradiation in some borate and silicate glasses导致了positive hole centers在类似位置(410 to 500 nm)的吸收。
(3) 飞秒激光还原的机理:It is well-known that femtosecond laser pulses can create electrons and holes in the materials throughout multiphoton excitation. The irradiation of the glass by femtosecond laser creates electron-hole pairs by band-to-band transition via multiphoton excitation. This means that the electrons are delocalized from the valence band (VB) and transferred to the conduction band (CB). The electrons and holes are redistributed throughout the conduction and valence bands. As a result, some electrons were trapped by the Eu3+ ions which led to the formation of the Eu2+ ions. Some holes were trapped by the trapped centers that gave rise to the appearance of the positive hole centers which induced unique absorption in the range from 400 to 500 nm.
飞秒激光的应用:femtosecond pulses provide a promising technique to fabricate optical device for potential applications such as optical waveguides, waveguide amplifiers, and 3D optical memory.
基于变价的3D信息存储:One potential method of the fabricating 3D optical memory is the valence manipulation of samarium ions in micrometer-scale dimension inside a transparent glass with femtosecond laser irradiation pulses, where the data information can be read out in the form of luminescence.
3. Sm3+掺杂铝硼酸钠玻璃
[Valence state manipulation of Sm3+ ions via a phase-shaped femtosecond laser field-Photonics Research-2018]
4. Bi3+掺杂介孔石英玻璃+飞秒激光
[Space-selective control of luminescence inside the Bi-doped mesoporous silica glass by a femtosecond laser-JMC-2009]
Bi3+在800 nm飞秒激光辐照下,部分地变为Bi2+或Bi+,而材料本身是不吸收800 nm (non-resonant wavelength)光的,所以这个photoreduction是非线性过程。It is suggested that the multiphoton absorption process and photoreduction of Bi3+ ions are involved in the experimental phenomenon. Through Joule heating, multiphoton ionization, and collisional ionization, free electrons are created in the fs laser-focused area. After that, Bi3+ ions capture the free electrons and hence transform into Bi2+, and even Bi+ ions. The holes would be trapped at nonbridging oxygens in the SiO4 polyhedrons. Note that Bi3+ + e− → Bi2+ and Bi2+ + e− → Bi+ are very similar to the conventional chemical processes. It is suggested that control of the laser parameters can tune the active centers for the luminescence tuning of main group metal ion-doped phosphors.
5. Sm3+掺杂钠铝硼酸盐
[Permanent photoreduction of Sm3+ to Sm2+ inside a sodium aluminoborate glass by an infrared femtosecond pulsed laser-APL-1999]
(1) Recently, it was found that direct excitation of the CTS and 5d – 4 f transition bands can induce photoreduction in some rare-earth ions.(溶液中)
(2) 800 nm fs pulsed laser辐照后吸收谱的变化:通过对比还原气氛下得到的Sm2+掺杂玻璃的吸收谱,醉着认为吸收谱的差异对应于辐照后得到的Sm2+的吸收。
(3) Sm3+在700-900 nm范围没有吸收,所以光还原是一个多光子还原的过程。
(4) EPR:对于没有辐照的样品,没有看到apparent signals,但是对于辐照后的样品,出现了一个在g =2.0的峰,可以归属于defect centers of holes trapped by nonbridging oxygen ions and tetrahedral coordinated boron atoms, and electrons trapped by the quasi-F centers as in gamma-ray-irradiated sodium borate glasses.
(5) 机理:Following the irradiation of the focused femtosecond laser, active electrons and holes were created in the glass through multiphoton ionization, Joule heating, and collisional ionization processes. Holes were trapped by nonbridging oxygen ions as well as by tetrahedral coordinated boron atoms, while some of the electrons were trapped by the Sm3+.
(6) 热稳定性:The trap levels of defect centers may be deep, resulting in stable Sm2+ at room temperature. 作者做实验研究了光还原得到的Sm2+的热稳定性,发现室温的时候Sm2+很稳定,但是一旦温度超过Tg(玻璃转化温度),就被氧化为Sm3+。作者这里并没有说明thermal bleaching的气氛。
日本
日本-固体
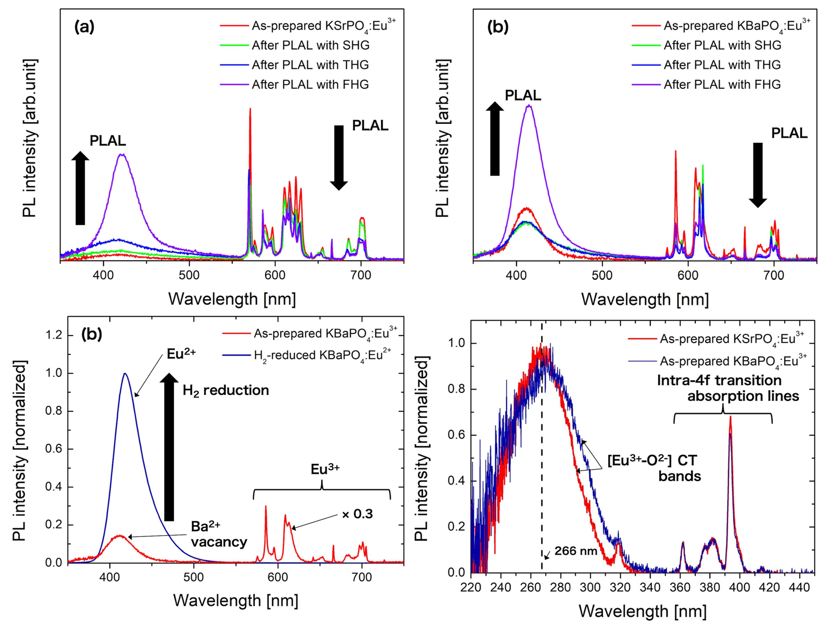
1. KSrPO4:Eu和KBaPO4:Eu
[Micronization of KSrPO4:Eu and KBaPO4:Eu phosphor particles for white light-emitting diodes by pulsed laser ablation in liquid-Optics & Laser Technology-2021]
- PLAL caused not only the micronization but also the reduction of Eu ions.
- SHG是second harmonic generation of Nd:YAG,对应于532 nm,THG是三阶倍频对应355 nm,FHG四阶倍频对应266 nm。266 nm的最有效。
- 对KBaPO4来说,SHG和THG似乎还让Eu2+发光变弱了。
- Considering the PLE spectra and the laser wavelength dependence of reduction by the photo excitation, the photon energy of wavelength corresponding to [Eu3+ − O2−] CT is probably the threshold to induce the reduction by the photo excitation.
- 还原过程:
(1) 由于电荷迁移态Eu2+ (Eu3+ + e−) and O− (O2− + h+)。However, they usually recombine to their original state and release the energy as the luminescence at the same time. Therefore, the hole generated at O2− must be trapped somewhere in order to stabilize Eu ions in divalent. PO4 groups in phosphate phosphor can probably trap the hole because SiO4 groups in Sr2SiO4:Eu are reported to be able to trap the hole.
(2) Sr或者Ba的空位捕获了hole,作者观察到了Ba和Sr空位的发光(和还原气氛的Eu2+发射波长不同),但是没有直接证据。(我:可能是得到占据不同格位的Eu2+) - 激光作用范围:In the case of the reduction by the photo excitation, only Eu ions existing near the particle surface within a penetration depth of pulsed laser are reduced unlike in the case of H2 reduction process.
2. KSrPO4:Eu
[Effect of laser fluence and charge compensation defects on photoreduction of Eu ions in KSrPO4 crystal using a UV laser-JL-2022]
3. SrAl2O4:Eu3+
[Radiation-induced reduction of Eu3+ doped in SrAl2O4 — Materials Letters - 2021]
Ionizing radiation的坏处:it may cause a risk to harm biological cells as it damages DNA.
Ionizing radiation的计量:One of the techniques is to monitor and record the dose history of radiation workers by using a so-called dosimeter. In a dosimeter, a luminescent material is used as a sensing element, which typically shows thermoluminescence (TL) , optically stimulated luminescence (OSL), or radio-photoluminescence (RPL) phenomenon.
PRL材料的典型代表:Ag-doped phosphate glass
本文用X射线辐照样品,然后热退,最高退到450度。
4. [Room-temperature persistent spectral hole burning of x-ray-irradiated Sm3+-doped glass—邱建荣-2000]
Emission-color change in Eu-doped high-symmetry glaserite-type silicates
Emission-color change caused by photoredox reactions of europium ions in Ba3MgSi2O8
5. Ca2ZnSi2O7:Eu2+
[Structural and photo-induced properties of Eu2+-doped Ca2ZnSi2O7: A red phosphor for white light generation by blue ray excitation-JL-2007]
[Photo-induced charge state conversion of Eu2+ in Ca2ZnSi2O7—JAP-2009]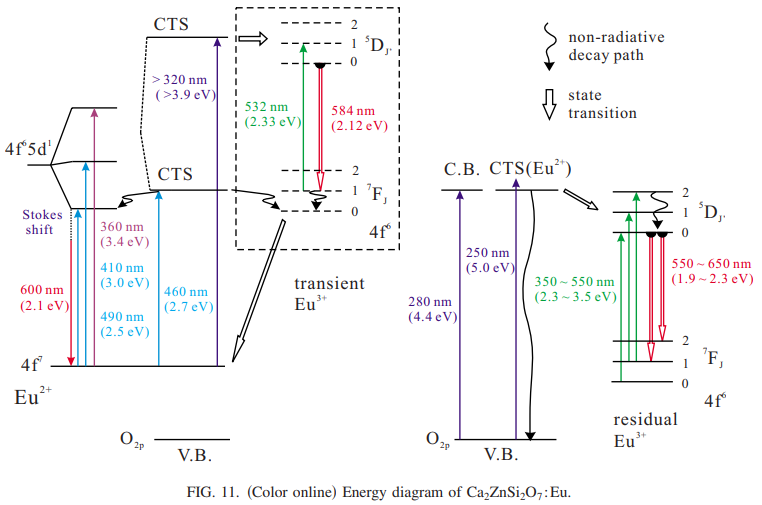 合成的是Eu2+的样品,但是通过瞬态光谱观察到了Eu3+的发光。The formation of a photo-induced transient Eu3+ state from Eu2+ was observed by the irradiation of UV light with an energy being slightly larger than the band gap energy of the host. A recovery life time of the Eu3+ to Eu2+ ions is evaluated to be 11.0 ms at 77 K by a pump-probe technique.
合成的是Eu2+的样品,但是通过瞬态光谱观察到了Eu3+的发光。The formation of a photo-induced transient Eu3+ state from Eu2+ was observed by the irradiation of UV light with an energy being slightly larger than the band gap energy of the host. A recovery life time of the Eu3+ to Eu2+ ions is evaluated to be 11.0 ms at 77 K by a pump-probe technique.
The pump-probe measurement verifies that the excitations above the band gap energy induce the formation of the transient Eu3+ ions with a recovery life time of 11.0 ms.
6. Eu3+ acting as electron trap in glass
[Eu3+ Ion as Electron Trap in Glass-Journal of the Physical Society of Japan-1967]
[Color Centers in X-Irradiated Soda-Silica Glasses-JACS-1964]

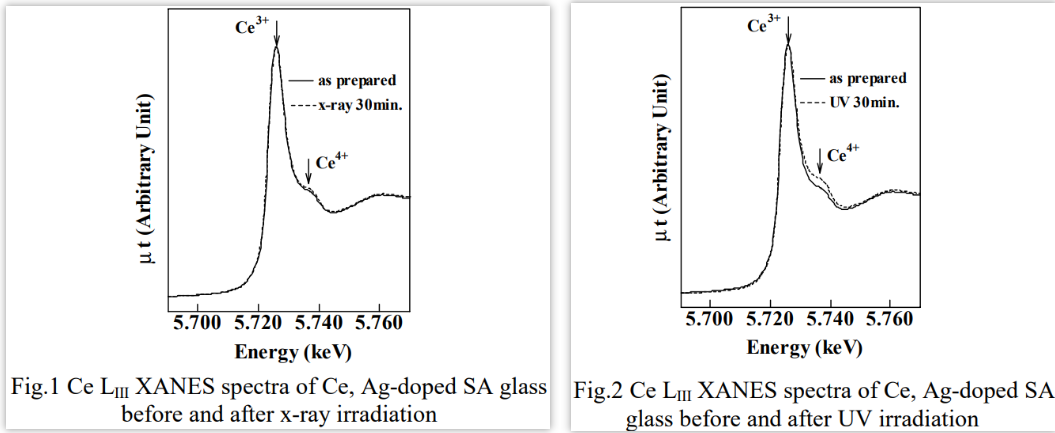
7. [X-ray and UV irradiation effects on Ce3+ ion doped in UV sensitive glass-1999]
When UV radiation irradiated on this glass, Ag+ ion is reduced to Ag atom. The role of Ce3+ ion in this glass is considered to be a sensitizer which sensitize the reduction of Ag+ ion.
7. [Excitation into Charge Transfer Band of Y2O2S:Eu-JES-1981]重点
9. [Reversible photoinduced spectral change in Eu2O3 at room temperature-APL-2001]
12. [Unique photoluminescence degradation/recovery phenomena in trivalent ion-activated phosphors-2015]
Possible applications of FEL in UV photochemistry and nuclear waste reprocessing-1993
日本-溶液
1. EuCl3 in organic solvent (photochemcial reduction)
[One-Electron Reduction of Eu3+ Ions Induced by the Irradiation of γ-Ray or UV-Light. The Fluorescence Properties of Eu2+ Ions in an Ethanol Matrix-Chemistry Letters-1988]
稀土在溶液中发光弱的原因以及改进的方法:However, the fluorescences of these ions are very weak in a solution because of a vibrational interaction between an excited Eu-ion and solvent molecules. Recently, it has been reported that the complexation of Eu2+ ions with crown ethers (冠醚) or cryptands (穴状配体) enormously increases the fluorescence intensity and the effect was due mainly to the protection of Eu2+ from the collision of methanol molecules by coordinating ligands and to the removal of the solvating OH group from Eu2+.
内容:In this paper, we report the one electron reduction of Eu3+ ions by the irradiation of γ-ray or UV-light in a low temperature ethanol matrix and the fluorescence properties of Eu2+ ions produced by these processes.
吸收谱的变化:The color of the solution was changed to purple after the γ-irradiation due to the trapped electrons formed. Upon photobreaching with a light>600 nm, new absorption bands with max at 260nm and 325nm were formed (Fig. 1. 2) which
2. Eu3+掺杂aqueous solution和alcoholic solution (Eu(ClO4)3)
[Higher yield of photoreduction from Eu3+ to Eu2+ with shorter wavelength irradiation-CPL-1992]
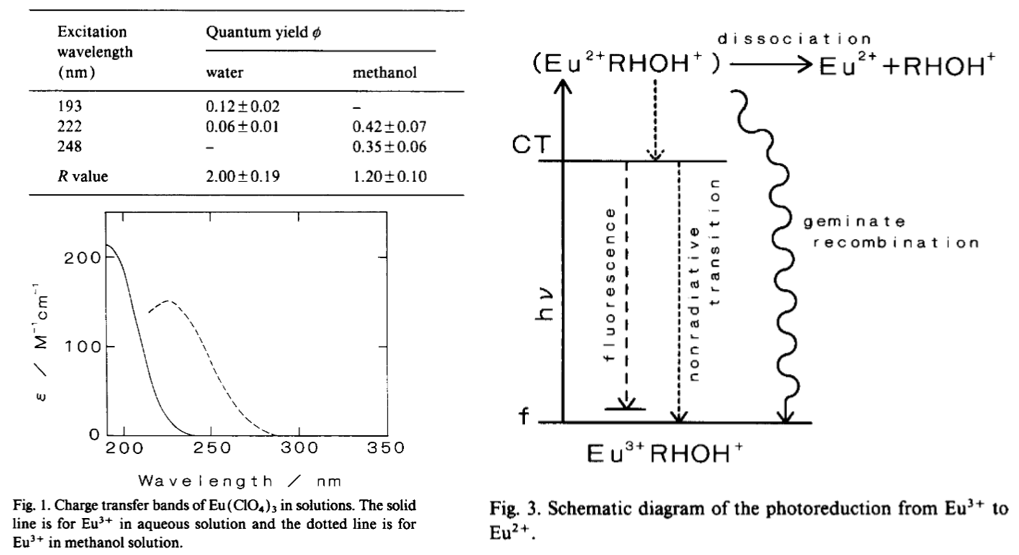
(1) 如果只激发f-f的可见光区,那么只有PL,没有redox reaction。Photoredox reactions have been known to occur when a charge transfer band, in other words, a 5d←4f transition in the UV region, is excited。
(2) Eu2+稳定性:溶液中光还原得到的Eu2+不稳定(所以测瞬态吸收谱):Although Eu2+ is not unstable in an aqueous solution, flash photolysis is a convenient means of measuring it before the second reactions (discussed later) and its eventual diffusional escape from the observing volum.
(3) 波长依赖光还原:作者只选取了两个波长,分别对aqueous solution和alcoholic solution的样品进行测试,计算四种情形下的光还原的quantum yield,发现都是短波长更有效。
(4) 波长依赖光还原的原因:激发CT态,形成了molecule Eu2+RHOH+,然后弛豫到发射能级,但是对应的fluorescence的QY很低。大部分的Eu2+RHOH+会存在一个dissociation和recombination的过程(两个过程竞争),前者对应于光还原。
(5) 光还原的quantum yield计算:利用消光系数还有参比物,计算光还原的效率。
3.Eu3+掺杂aqueous solution和alcoholic solution
[High photoreduction yield of Eu3+ to Eu2+ in alcoholic solutions and its wavelength dependence-JAC-1993]
(1) 应用:Photoredox reactions of lanthanide and actinide ions are expected to be utilized for nuclear fuel reprocessing.
(2) 辐照源:193 nm (ArF)、222 nm (KrCl)、248 nm ((KrF)、308 nm ((XeCl)
(3) alcoholic solution中Eu3+更容易被光还原,相比水溶液。
(4) 概述:The photoreduction yield of Eu3+ to Eu2+ depends on the kind of Eu3+ counter-ions and solvents and on the excitation wavelengths. The photoreduction yield can be explained in terms of the dissociation efficiency of the geminate pair (Eu2+ and oxidized ligand). Similar chemistry may be observed for other lanthanide as well as actinide ions in solution.
4.Sm3+掺杂methanol solution
[One-photon reduction of Sm3+ to Sm2+-Journalof Photochemistryand PhotobiologyA: Chemist-1997 ]

对于Ln3+来说,直接激发f-f跃迁的单光子过程只会导致PL,而不会有光还原,但是直接激发f-f跃迁的双光子过程可以导致光还原。已经有人报道激发CT或者f-d跃迁可以实现光还原,包括Ce3+/Eu3+/Sm3+。之前别人在溶液中实现了Sm3+的还原,但是认为这是一个多光子过程,但是作者在这篇文章中通过瞬态吸收光谱(nanosecond laser flash)证实这是一个单光子过程。The linear dependence of the concentration of Sm2+ on the laser fluence indicates that the photoreduction of Sm3+ to Sm2+ in solution occurs via a one-photon process
(1) 在甲醇溶液中,Sm3+到Cl-的CTB在220 nm有最大值。作者这里用的是KrF excimer laser波长为248 nm,和CTB匹配。
(2) 作者似乎并没有说光还原之后的Sm2+是否稳定,应该是不稳定,就像之前Eu2+。
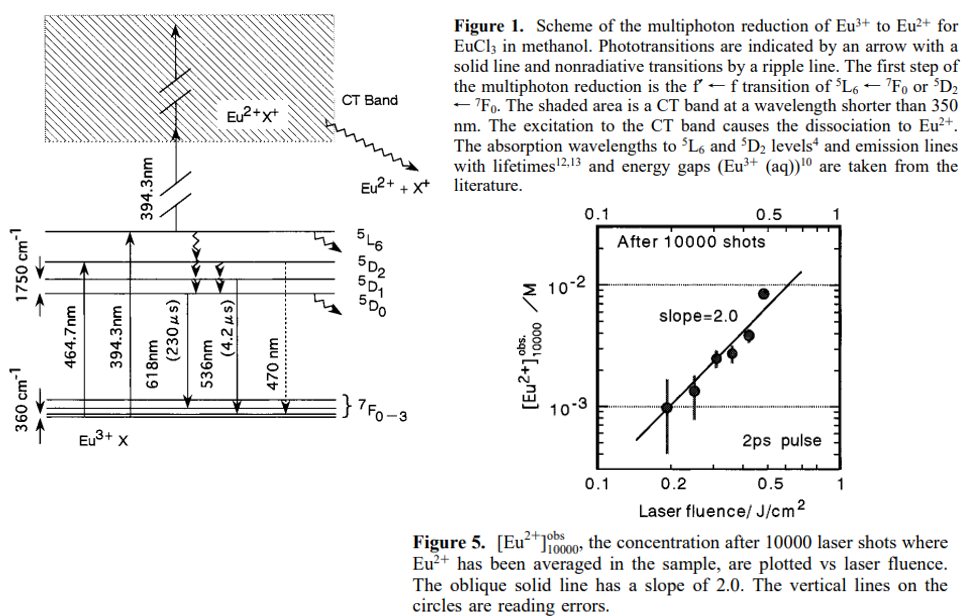
摘要:Multiphoton reduction of Eu3+ to Eu2+ in methanol was induced by irradiation with the second harmonic (394 nm, 2 ps) of a Ti:sapphire laser. The conversion efficiencies were increased to 0.3 for short pulse excitation, as compared to on the order of 10-5 for a nanosecond dye laser pulse. It appears that a three-photon process pumps a deep charge-transfer state from which Eu3+ efficiently dissociates to Eu2+. On the basis of the excitation spectra, the first intermediate state is the 5L6 state, which is one of the f-electronic excited states. The Eu2+ concentration increased shot by shot and approached a plateau. The saturation behavior will be explained in terms of the back photooxidation of Eu2+ to Eu3+.
6. EuCl3 in in methanol in the presence of 15-crown-5-ether
[Reduction of Eu3+ to Eu2+ by an intense femtosecond laser pulse in solution-CPL-2008]
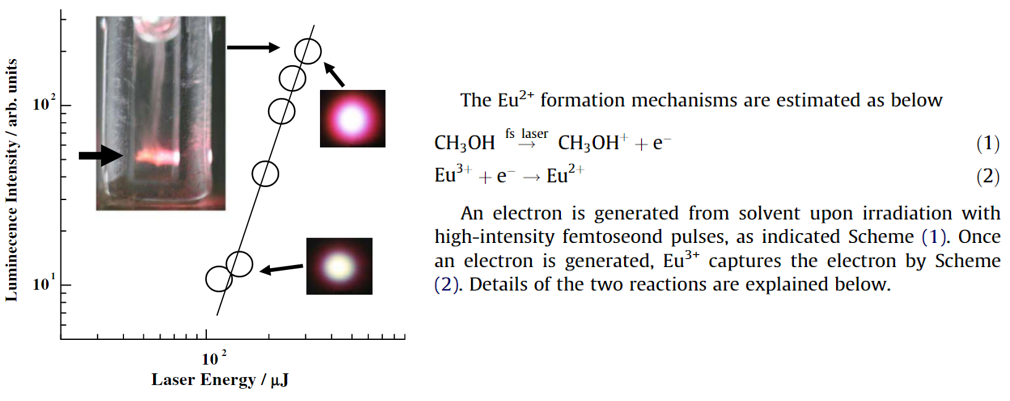
- White-light laser:the femtosecond white-light laser was generated during the irradiation.
- 摘要:Europium 3+ ions in methanol were found to be reduced to the corresponding 2+ ions upon irradiation with intense femtosecond laser pulses. The excitation wavelength of 800 nm was nonresonant with their electronic transitions of Eu3+. It is notable that femtosecond white-light laser was generated when the reactions occurred. The mechanisms can be explained in terms of solvated electron formation followed by the reduction. The electron ejection in a focused beam in solution has been known to be accompanied by white-light laser.
- 上图:The Eu2+ fluorescence intensity increases nonlinearly with increases in the irradiation energy. The solid line of the log–log plots has a slope of 2.9. The two right inserted figures are pictures of white-light laser on a paper 20 cm after laser passing through the sample cell for the cases of the highest and lowest irradiation energies. The size of the central white portion is approximately 40 mm in diameter. When the Eu2+ fluorescence was observed, the femtosecond white-light laser was emitted. The top picture is a side view of the cell, showing luminescence and bubbles during irradiation. The black arrow on the picture indicates the incident laser direction.
7. SmCl3 in in methanol in the presence of 15-crown-5-ether
[Reduction of Sm3+ to Sm2+ by an Intense Femtosecond Laser Pulse in Solution-JPCA-2010]
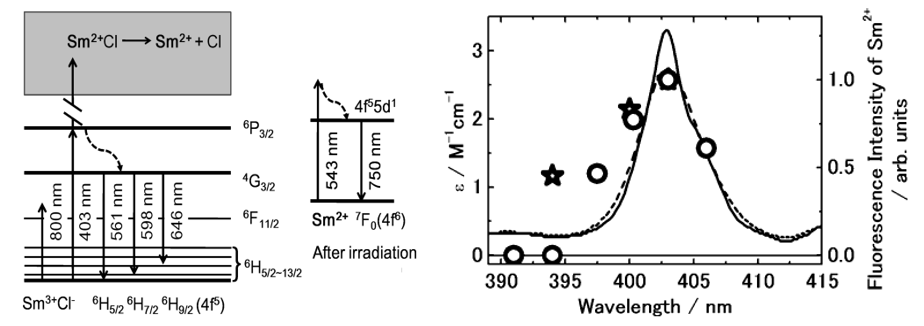
- 4f electronic excited states反应活性低的原因:The lanthanide ions have 4f-4f (ff) transitions in UV-vis-IR regions with atomic-like line spectra because f-orbital electrons have high electron densities near the nuclei and have weak interactions with the surrounding solvent. As a result, the 4f electronic excited states show low reaction activities; in fact, no photoredox reaction has been reported from the lowest emissive states in the 4f electronic excited state in solution.
- 三种方法实现Eu3+的photo-reduction:
-
-
- CT state: excitation to the CT state by one-photon absorption, and the reaction quantum yields are determined to be relatively high (0.1-0.9) dependent on the solvent and the excitation wavelengths.
- resonant multiphoton reaction:The first laser light was tuned to one of the ff transitions (394 nm, from 7F0 to 5L6), and the second and/or third photon was pumped up to the CT states using nano- and picosecond laser pulses. One of the important findings is that a short duration pulse excitation dramatically improves the reaction efficiency.
- 飞秒激光(应该也是multiphoton reaction,不过是nonresonant multiphoton reaction):focused femtosecond laser pulses at a nonresonant wavelength. The scheme is explained in terms of a reduction to Eu2+ in response to solvent ionization, followed by electron capture by Eu3+.
-
- Sm2+ in in methanol in the presence of 15-crown-5-ether有宽带发光(750 nm),543 nm可以有效激发Sm2+到4f55d1态。通过检测Sm2+的发光强度,可以反映Sm2+的生成量。
- 403 nm levels work as intermediates of the multiphoton reaction.
- 飞秒实验:作者这里也采用了非共振的飞秒激光来还原Sm3+,作者认为其机理和邱建荣的类似。
- 上面右侧图中偏移是因为光的波长不纯。
-
8. Yb3+ (YbCl3·6H2O) in MeOH and EtOH
[Reduction of Yb(III) to Yb(II) by Two-Color Two-Photon Excitation-JPCA-2013]

- Absorption spectrum of Yb2+ significantly depends on solvents. The absorption and emission spectra of Ln2+ (Ln = Eu or Sm) are known to be sensitive to solvents. 作者在室温下并没有观察到Yb2+的发光,虽然有明显的吸收。
- 电荷迁移态:The CT levels for YbCl3·6H2O in MeOH and EtOH have peaks at 210 and 240−245 nm and had a tail that extended over 270 nm; therefore, the tail can be excited by the fourth harmonics of the Nd3+:YAG laser (266 nm).
- 还原氧化的双重作用:266 nm开始的时候可以还原Yb3+,但是Yb2+在266 nm或者355 nm处也有吸收,所以266 nm也可以氧化Yb2+。因此用266 nm的光作用在初始样品上,会观察到Yb2+随着辐照增加而增加,随后达到平衡状态,即单位时间还原得到的Yb2+ = 单位时间氧化得到的Yb3+。
- 在近红外区,作者观察到了很弱的Yb3+的降低,说明Yb2+的出现是以消耗部分Yb3+作为代价的。
- 波长依赖实验:[Higher yield of photoreduction from Eu3+ to Eu2+ with shorter wavelength irradiation-CPL-1992]报道了波长依赖的UV light-induced photo-reduction,发现波长越短越有效。对于作者的实验来说,The CT state with higher energy may give higher reduction yields for this Yb3+ system, but it is not clear at the present stage. To discuss precisely the UV wavelength dependency, the present experimental errors were too large.
- 误差来源:would contain several tens of percent uncertainty due to the lack of precise measurements of the focused beam size and intensity distributions in the spot。
9. Eu3+ in aerated water in the presence of 2-propanol.
[Reduction and precipitation of aqueous europium (III) under an air atmosphere by near-infrared femtosecond laser pulses-2022]

- 第一种方法:reduce Eu3+ by exciting the charge-transfer (CT) band in degassed alcohol with nanosecond laser pulses of VUV or UV wavelength. This procedure has been shown to be effective for not only lanthanoid ions but also noble metal ions dissolved in radioactive waste solution.
- 第二种方法:resonant two-photon excitation to CT state via the f-f transition using visible laser pulses. We have found two photon reduction of Eu3+ to Eu2+ in nano- and picosecond pulse excitations. The reduction efficiencies of Eu3+ were largely improved from on the order of 10−5 to 1 by picosecond pulse excitation. The improvement would be largely reinterpreted by the simple reasons of pulse shortening. The laser intensity should have increased 1 × 104 times by the shortening pulses from 20 ns to 2 ps under the same focusing conditions.
- 第三种方法:reduction of lanthanoid ions with electrons ejected from solvent by nonresonant multiphoton ionization using focused near-infrared (NIR) femtosecond laser pulses. Lanthanoid ions were successfully reduced in degassed alcohol using this approach.
- Highlights:
(1) Near-infrared femtosecond laser pulses cause nonresonant multiphoton ionization of water.
(2) Trivalent europium is reduced by hydrated electrons generated in aerated water in the presence of 2-propanol.
(3) Precipitation of europium sulfate is investigated by absorption/extinction spectroscopy and transmittance measurements. - 结论部分:This work reveals that the reactive species generated in water by femtosecond laser pulses can reduce europium ions even under an aerated condition. The absence of oxygen is not the critical factor, but hydroxyl radical scavengers are indispensable to achieve the reduction of europium ions.
注:Hydroxyl radicals are highly reactive species that attack most of the organic molecules. They are highly oxidizing in nature which is attributed to their oxidation potential
10.
[An electron-capture efficiency in femtosecond filamentation-CPL-2022]
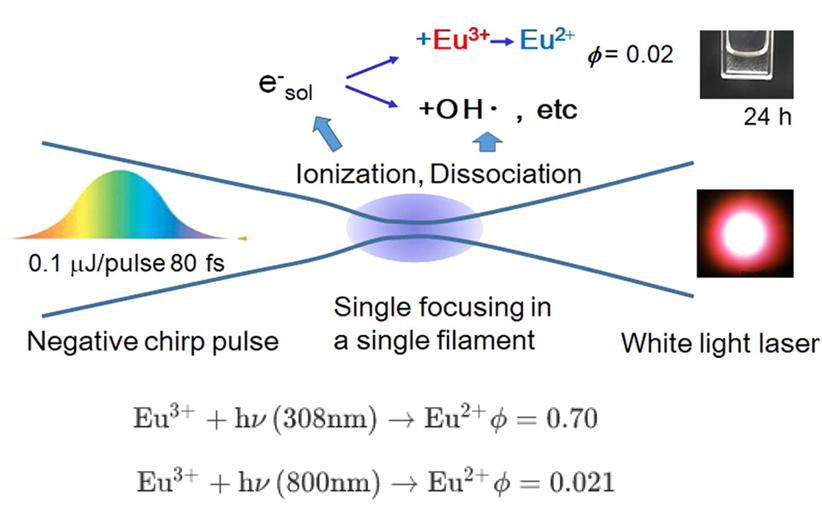
- The single UV photon reaction showed a tendency toward saturation in the product absorbance of Eu2+ as the number of laser shots. This was simply because of the back photo-reaction of and an approach to a photo-stationary equilibrium. The back reaction was in fact observed with a quantum yield of 0.35 at 308 nm laser irradiation starting from a fresh Eu2+ sample.
韩国

- Sm2+ ions show a high persistent spectral hole burning efficiency. 但是一般来说Sm3+更稳定,所以it is thus needed to reduce Sm3+ to Sm2+ for use PSHB in frequency domain optical storage.
- 空气中Sm3+自还原的基质BaB8O13 or SrB4O7.
- 存在不同的方法得到Sm2+,不同的方法可能导致不同的光学特性,比如photo-bleaching effect。本文repared SrB4O7 and SrB6O10 crystals by using two different reduction methods, i.e., thermal treatment in air and X-ray irradiation.
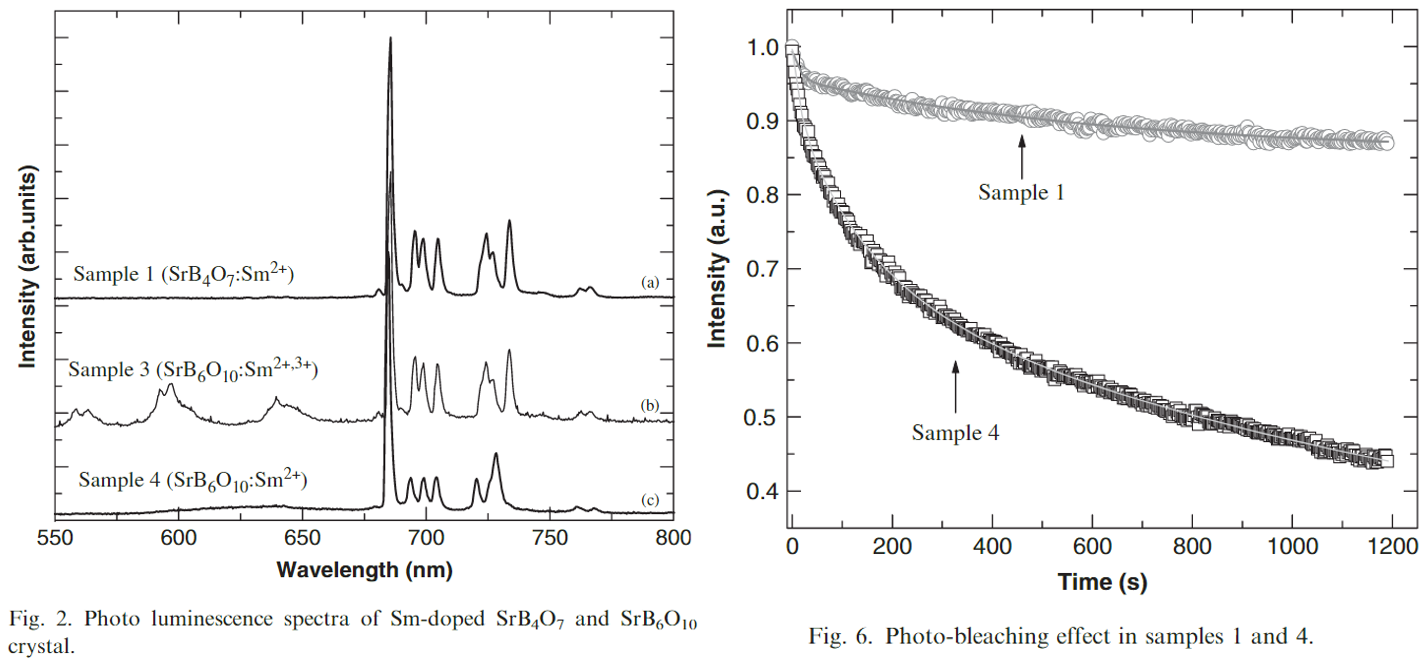
- xx
2. [Conversion of the valence states of Eu ions in YVO4 with the gamma-ray irradiation-2016]
中国
黄彦林
中文论文:
1.BaBPO5:Sm3+
[Structural defects and luminescence properties of Sm2+ ions doped in BaBPO5 phosphor by X-ray irradiation-JAC-2009]
(1) 激发谱的差异:不同方法制备出来的Sm2+掺杂BaBPO5的发光特点不同,作者这里是通过X射线辐照得到Sm2+,别人通过还原气氛得到的Sm2+,激发谱有些许差异。This difference may be due to the different preparation method.The X-ray-irradiated sample may have different defect structures compared to those samples heated in H2-gas.
(2) 热稳定性:To check the thermal stability of Sm2+ ions, the X-ray-irradiated sample was heated at several temperatures in air and then rapidly cooled down to room temperature. Then the emission spectra were measured at RT and are shown. It is found that the Sm2+ ions were stable up to a temperature of 450 °C. At the temperature 250 °C, the intensity of Sm2+ emission keeps stable. At the temperature from 250 to 450 °C, the intensity of Sm2+ fluorescence decreased. This could be understood that the Sm2+ ions reduced by X-ray irradiation were oxidized by heat treatment in air. The oxidation of Sm2+ was constantly increased with increasing the thermal treatment temperature. It can be explained that the electrons or holes in the trapped center are released by thermal energy and then recombined with Sm2+ ions resulting in the Sm3+ ions.
2. Ba2SiO4:Sm3+
[The Irradiation Induced Valence Changes and the Luminescence Properties of Samarium Ions in Ba2SiO4-JES-2010]

- Sm2+也可以作为probe,就像Eu3+,也存在\({ }^{5} \mathrm{D}_{0} \rightarrow{ }^{7} \mathrm{~F}_{0}\)的nondegenerate的emission。变温实验发现了存在三种Sm3+ crystallographic sites have different temporal and spectral behavior from each other,但是只有一种是在室温下能发光的。
- 488 nm是常用的实现Sm2+的photobleaching的激光波长(光一方面减少Sm2+,一方面激发Sm2+使其发光),Sm2+发光强度和激光的作用时间可以用一个双指数来拟合,This indicates that there are at least two kinds of decay times during the photobleaching.
- It is well known that, under X-ray irradiation, the O components are easy to be ionized to release an electron that could be captured at Sm3+ sites, which results in the reduction of Sm2+.
- Actually, the real mechanism of photobleaching effect and hole burning of Sm2+ ions are not clear at present; however, it can be widely accepted that the photobleaching process is charge transfers from the Sm2+ ions to nearby defects.
- The photobleaching experiments show that the luminescence intensity decreases quickly as the 488-nm laser irradiation time is increased. Usually, this photobleaching effect is ascribed to the photoionization of Sm2+ to Sm3+. This shows that the Sm2+ ions formed by X-ray irradiation are not stable. It is reasonable to say that these defects induced by X-ray irradiation play an important role in the photobleaching.
3. Sm3+掺杂Li2O–SrO–B2O3玻璃
[Huang Y, Jiang C, Jang K, et al. Luminescence and microstructure of Sm2+ ions reduced by x-ray irradiation in Li2O–SrO–B2O3 glass-JAP-2008]
(1) 光学烧孔:Luminescence of Sm2+ has been paid an intense attention because this ion is known to have a property of persistent spectral hole burning (PSHB). Sm2+掺杂玻璃对PSHB来说很有吸引力,因为glasses are the wellknown host in which the inhomogeneous linewidth is significantly broadened,而且, PSHB observed in glass matrices containing Sm2+ ions showed relatively high thermal stability。之前也有报道通过X射线辐照得到的Sm2+掺杂玻璃表现出比还原其分析得到的样品更好的光学烧孔的性能。
(2) 5D0→7F0 (Sm2+):两个能级都是非简并的,所以不能在CF下劈裂,因此图中该峰的宽化reflect the bonding environment of the Sm2+ ions with large inhomogeneities.
(3) 弱宽峰PL:可能来自Sm2+的d-f跃迁,也可能是来自defect centers的发射。
4. The luminescence and stabilities of Sm2+ ions doped in LiBaB9O15
5. Radiation-Induced Effects and Valence Changes of Samarium Ions in Sr3Sm ( PO4 ) 3
5. Temperature-Dependent 5D0-7F0 Luminescence of Sm21 Ions Doped in Alkaline Earth Borophosphate Glass
非黄彦林
1.AOM综述
[Tuning the Luminescence of Phosphors: Beyond Conventional Chemical Method-AOM-综述-2015]
光辐照 versus Ionizing Radiation
Photoreduction of active ions with ultrashort-pulse lasers is one remarkable way to modulate the luminescence of phosphors. Once the valence state of the activator centre changes, the luminescence of phosphors will be modulated.
Compared to photons, ionizing radiation possesses much higher energy. Ionizing radiation is capable of liberating electrons from atoms or molecules and breaking chemical bonds, thereby ionizing them. Utilizing ionizing radiation with appropriate doses, the luminescence of phosphors can be modified due to the radiation-induced defects or radiation-induced tuning of dopant valence states, or radiation-induced matrix phase evolution.
4.3.1 Femtosecond Laser-Induced Tuning
2. SrAl2O4:Eu2+和Y2O3:Eu3+
[Photoluminescence and gamma-ray irradiation of SrAl2O4:Eu2+ and Y2O3:Eu3+ phosphors-JPCS-2007]
In irradiated samples, the brightness is decreased without sensible change in the wavelength distribution of the luminescence spectrum and in the decay kinetic upon gamma exposure. Moreover, the emission due to Eu3+→Eu2+ conversion in Y2O3:Eu3+ phosphors was not observed in our sample after irradiation to high exposure. Also the brightness of SrAl2O4:Eu2+ phosphor turned out to decrease after the exposition to ionizing radiation while the luminescence wavelength distribution remained unchanged.
可能的原因:the observed lowering of the brightness could be explained by some kind of radiation-related process, which lowers the concentration of emitting ions. Another possible explanation could be the radiation-induced quenching of some emitting ions: in fact, the quenching could be so fast that it cannot be detected by analyzing the decay kinetic curve of the overall system. As far as Eu2+ samples are concerned, we think the holes and electrons generated by gamma irradiation can recombine and trap at Eu2+ site, which may play an important role in leaving unchanged the color coordinate of the phosphors. Further study is necessary to better understand and prevent the reduction of relative brightness of analyzed phosphors.
3. 氟化镁锶中铕和铽的电荷迁移及其平衡解析-物理化学学报—1995
4. Y2O3:Eu3+
[Ultraviolet light-induced spectral change in cubic nanocrystalline Y2O3:Eu3+-CPL-2003]
6. [The mechanism of photobleaching in Sm2+-doped alkaline-earth fluorohalides-JL-2005]
7. [Photo-Bleaching Effect in Divalent Samarium-Doped Alkaline-Earth Fluorohalides—2004]